Have you ever wondered how the detergent that you put in your washing machine gets rid of the stains on your clothes? It may seem like an incredible phenomenon that one might associate with magic. However, it is clearly a scientific process. Firstly, you need to understand that there are two different types of molecules. Molecules are classified as either covalent or ionic. Secondly, the fundamental difference between these two classes is that covalent molecules are made up entirely of nonmetal atoms that share electrical charges while ionic molecules are made up of both metal and non-metal atoms. The covalent class can be divided further into two subclasses, nonpolar covalent and polar covalent.
Thirdly, a nonpolar covalent molecule emits no electrical charge whereas a polar covalent molecule emits an electrical charge. Polar covalent compounds and nonpolar covalent compounds cannot dissolve each other. For example, water is a polar covalent compound and vegetable oil is a nonpolar covalent compound. When you put vegetable oil and water together, they don`t mix. The water remains on the bottom while the oil goes to the top. However, polar covalent compounds can dissolve other polar covalent compounds and also ionic compounds because both compounds emit an electrical charge. Salt, which is a polar covalent compound, can be dissolved in water, which is also polar. Nonpolar covalent compounds, on the other hand, can only dissolve other nonpolar covalent compounds.
[caption id=“attachment_9633” align=“alignnone” width=“480”]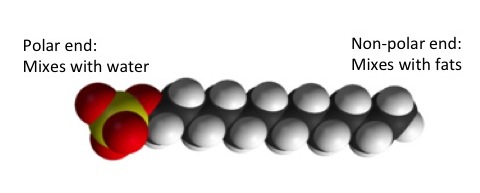 via soapbubble.dk[/caption]
via soapbubble.dk[/caption]
Washing detergent is made up of complex molecules that are ionic on one end and nonpolar on the other. The nonpolar side of a soap molecule latches onto and dissolves the stains that are present in the clothes that one puts in the washer, like grass or blood stains. Suddenly the ionic side of the molecule is attracted to the water, which is a polar covalent compound. Thus, the entire molecule is removed from the clothing, bringing the stain`s molecules with it, while dissolving in the water. Voila! Your clothes are now stain free. But if you have a stubborn stain that won`t come off your clothes, your first thought may be to pretreat the stain before washing. When you pretreat stains on your clothes, all you are doing is adding a concentration of soap directly onto the stain. The intended purpose, therefore, is that the nonpolar covalent side of the molecules will dissolve the entire stain because you are putting so much soap directly onto the stain. Now you know how soap works at a molecular level.










